By Melissa Wallace
Through gene therapy, scientists in Alberta are offering a glimpse of hope to sufferers of a rare retinal disease
Choroideremia (CHM) once signalled utter despair among patients. Affecting 1 in 50,000 people – 90 per cent of whom are men – this rare, inherited disorder is caused by a genetic defect of the X chromosome which prevents the production of an important protein and eventually kills retinal cells. For people with choroideremia, this meant experiencing a gradual loss of vision starting in childhood and total blindness by the age of 40.
But a research team at the University of Alberta’s MacDonald Lab is hoping they’ve figured out how to stop the process.
In June 2015, the team pioneered Canada’s first human gene therapy trial for eyes with a mission to preserve and potentially restore vision for people with CHM. Six Canadians with CHM who had expressed interest in the clinical trial were selected from 20 individuals. They underwent surgery where a vector (or customized virus) was injected, transferring healthy, genetic material into the retina. Next, patients received regular monitoring and endured a series of tests to gauge the clinical effect of the vector. The trial concluded in June 2017 and the team is now pouring over the results.
“This disorder is slow and progressive, so we’re going to see these patients every year and monitor them to find out if the treatment has had an effect,” explains Dr. Ian MacDonald, team leader and Professor Emeritus at the University’s Department of Ophthalmology and Visual Sciences.
The MacDonald Lab includes three research bays, a tissue cultural room and a translational research base. Though much of the equipment tends to be fairly basic, such as thermocyclers for doing PCRs, the lab boasts a fairly extensive cell culture area for viral vector production and one piece of equipment that hadn’t been available in western Canada.
“The Microperimeter is a magnificent machine that would’ve been unavailable to us if we hadn’t undertaken this trial,” says MacDonald. The equipment, funded by the Canadian Foundation for Innovation and Alberta Advanced Education, analyzes the central visual field of the retina by tracking the eye, projecting targets onto the retina at specific points that can be periodically remeasured to determine change. Other organizations that have provided grants to the MacDonald Lab include The Canadian Institutes of Health Research (CIHR), Alberta Innovates, the Foundation Fighting Blindness and the Choroideremia Research Foundation, Canada Inc.
MacDonald first began focusing on choroideremia in 1983 while working in Ottawa. Patients and their families in the eastern Ontario region asked if he knew of anyone who was researching the disease. When he found no one was, he began a journey from gene mapping to gene cloning and then gene therapy.
In 1992, when work took him west to Edmonton, an experience with his neighbour provided additional motivation to continue his research.
“The fence between our properties needed repairs, but I didn’t want to get into that and antagonize the neighbour,” he says. “But he came along with his daughter to see me because she had a problem. Turns out, the daughter was a CHM carrier. Her father was tested and discovered he also had CHM and couldn’t even see the fence.”
CHM affects males significantly more than females because females have two X chromosomes. When one of the female’s chromosomes carries a faulty copy, the functioning copy will compensate. This means female carriers of CHM typically have mild symptoms and can usually retain good visual function throughout their lives.
MacDonald is still in touch with that neighbour and hopes CHM therapy will safely assist the six patients – all in their early 30s to early 40s – who underwent the trial. But they’re not the only ones eyeing a positive outcome. Patients have approached MacDonald about researching other disorders that could profit from gene therapy. If the CHM treatment is found to be effective, it could expand the potential to alleviate symptoms of other disorders such as Stargardt’s disease, Usher syndrome and achromatopsia.
“Gene therapy for some of these disorders has already occurred in animals and is now being undertaken in humans,” says MacDonald. “We don’t know exactly what the outcomes will be, but we perceive it will be favourable and maybe one day, gene therapies will be used for both rare and common conditions.”
As for a timeline of when they can deem the therapy a success, Chris St. Laurent, one of the lab’s research technicians, explains the top priority is to ensure the vector’s safety and that it will not cause further damage to the patients.
“This was a safety trial to see whether patients could tolerate the vector and if it could halt further progression in the degeneration of the disease over the next five or 10 years,” says St. Laurent. “That makes it difficult to tell if it’s working immediately, hence the need for such a long period of follow-up in these patients.
“But if there was no trial and these patients didn’t get any intervention, it would be a slow decline into blindness and there’s no way back from that.”
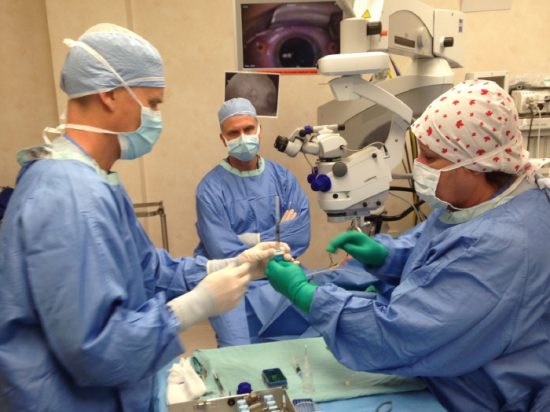
The team at MacDonald Lab continues to work collaboratively with Health Canada and Environment and Climate Change Canada.
“What is very important to Health Canada is that they have an example of a gene therapy trial in the human eye that they can benchmark,” says MacDonald. “They require an experimental protocol first and then a Health Canada application, whereas Environment and Climate Change Canada has to be involved because these are disabled viruses that are coming into our country.”
Another issue is cost. CHM is only the second disorder in which gene therapy in the eye has been undertaken, the first being Leber’s congenital amaurosis. Spark Therapeutics, a company based in Philadelphia, released Luxturna, an FDA-approved vector to cure this disease for $850,000 (treating both eyes).
“That’s an enormous figure and there will be challenges ahead for the health care system to bring these trials into the clinical domain, but it will happen,” says MacDonald.
Three other centres in the world have conducted similar CHM gene therapy trials and most are accumulating or finishing their trials now. A centre in England selected 30 CHM patients and Germany and Miami performed the therapy on six each.
“When asked whether they would choose to do this again, all of our patients said ‘yes,’” says MacDonald. “They’ve put their eye health on the line as an experiment and have been quite brave about it. We hope there will be some positive benefit for them.”
Cutline for image: (From left to right): The MacDonald Lab team members: Matthew Benson, Chris St. Laurent, Nicole Noel, Lance Doucette, Stephanie Hoang, Geoff Casey, and Dr. Ian MacDonald.
 BioLab Business Magazine Together, we reach farther into the Canadian Science community
BioLab Business Magazine Together, we reach farther into the Canadian Science community





