By Gabriela Martinez
It is recognized that environment is a potential source of contamination for food processing facilities. The pathogens, which occur naturally in the environment, can easily enter food processing areas several ways. Once inside, pathogens can be temporary visitors (present until they are removed via cleaning and disinfection) or they may persist in sites such as damaged areas of the floor or drains. Therefore, it becomes critical to set up and maintain an environmental monitoring program; particularly since the Food Safety Modernization Act (FSMA) is focusing on prevention. Food processors are more and more responsible for prevention of any adulteration in facilities and final product. Moreover, a good environmental monitoring program (EMP) allows companies to manage and possibly reduce operational, regulatory and branding reputation risks.
The setup of the EMP starts with identifying and documenting all physical areas which could be potential pathogen sources (raw materials, storage and shipping areas, etc.) and cross-contamination vectors (employees, equipment, pest, etc.). These areas and vectors should be surveyed, controlled and when possible, eliminated. Thus, the implementation of effective controls which includes microbiological sampling of high-risk areas will be part of the program. Sampling for pathogens or indicator microorganisms (see next page) in food contact areas during production is also vital. This offers confidence in the safety of the manufactured product. Additionally, the EMP helps to be aware of the plant environment and to measure the efficiency of the pathogen-prevention program. Furthermore, in an environmental monitoring program it is not only critical to test for pathogens, but also for the overall effectiveness of cleaning and sanitizing procedures. Both procedures are needed and both must be done properly to reduce microorganisms to safe levels. The goal of a cleaning process is to completely remove food and other types of soil from surfaces. Since soils vary widely in composition, no one detergent is capable of removing all types. In general, acid cleaners dissolve alkaline soils (minerals) and alkaline cleaners dissolve acid soils and food wastes. So, the personnel involved need to understand the nature of the soil to be removed before selecting a detergent or a cleaning regime. It is also essential that the cleaner matches with the water properties and is compatible (not corrosive, for example) with the surface characteristics on which it will be applied. However, not only the correct choice of agent is necessary for an optimal result; it should be coupled with a mechanical action, an appropriated contact time and correct operating temperature. As the combination of these parameters is characteristic to each process it becomes essential to verify effectiveness through sampling. Finally, cleaning is closely related to sanitation, because what hasn’t been previously cleaned can’t be sanitized.
The official definition (Association of Official Analytical Chemists) of sanitizing for food product contact surfaces is a process which reduces the contamination level by 99.999% (5 logs). Sanitation may be achieved using either heat (thermal treatment) or chemicals. Hot water sanitizing is commonly used where immersing the contact surfaces is practical (e.g., small parts, utensils). Hot water sanitizing is effective only when appropriate temperatures can be maintained for the appropriate period of time. For example, depending upon the application, sanitation may be achieved by immersing parts or utensils in 77 C to 85 C water for 45 seconds to five minutes. The advantages of this type of sanitation are: easy application, readily available, effective for a broad range of microorganisms, non-corrosive, and penetrates cracks and crevices. Nevertheless, it is relatively slow, can contribute to high energy costs, may contribute to the formation of biofilms and may shorten the life of certain equipment parts (e.g., seals and gaskets). Furthermore, fungal spores will survive this treatment.
Concerning chemicals, there is no perfect chemical sanitizer. Performance depends on: sanitizer concentration (too low or too high is ineffective), contact exposure time, temperature of the sanitizing solution (generally, 21 C to 38 C is considered optimal), pH of the water solution (each sanitizer has an optimal pH), water hardness and cleanliness of the surface. Indeed, some chemical sanitizers, such as chlorine, react with food and soil becoming less effective on surfaces that have not been properly cleaned.
Of course, the effectiveness of the plant’s sanitation practices must be verified to ensure that the production equipment and environment are indeed sanitary. Operators employ many kinds of verification, including physical and visual inspection methods, as part of ongoing environmental hygiene monitoring programs. Portable ATP bioluminescence systems are widely used by industry to obtain immediate results about the sanitary or unsanitary condition of food plant surfaces. ATP results should be followed up with more in-depth confirmation testing, such as indirect indicator tests and pathogen-specific tests. Indirect indicator tests are based on non-pathogenic microorganisms (Ex. coliform, fecal coliforms or total counts) that may be naturally present in food or in the same environment as a pathogen bacteria. These indicator organisms are used to assess the overall sanitation or environmental condition that may signal or indicate the potential presence of pathogens that may cause significant health risks to consumers. The principal advantages of using indicator organisms in an EMP are:
• the detection techniques are less expensive compared to those used for pathogens;
• indicator microorganisms are present in high number and a baseline can be easily established;
• indicator microorganisms are a valid representative of pathogens of concern since they survive under similar physical, chemical and nutrient conditions as the pathogen.
However, indicator organisms are not a substitute for testing for pathogens. A positive result indicates possible contamination and a risk of foodborne disease. It is recommended that samples be taken immediately before the production starts, just after cleaning and sanitation have been completed when information regarding cleaning and sanitation are required. However, when the sampling is carried out on surfaces previously exposed to chemical germicide treatment, appropriate neutralizers must be incorporated into the medium to preserve viability of the microbial cells.
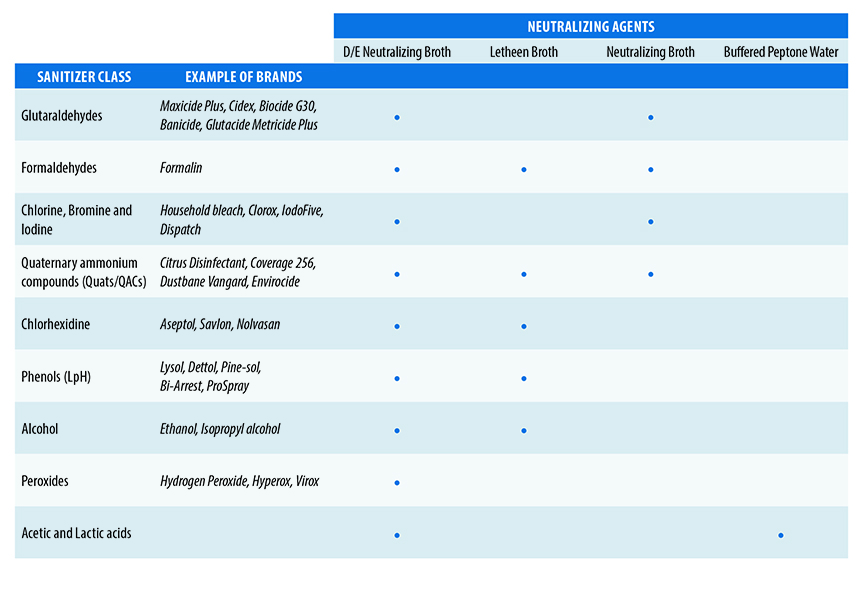
Neutralizers recommended for food plant monitoring include Dey-Engley neutralizing broth (DE), neutralizing buffer (NE), Buffered peptone water (BPW) and Letheen broth (LT). Most of them incorporated into a support such as a sponge, swab or chiffon to facilitate the neutralizing of residues of cleaning agents and sanitizers that may be picked up during swabbing.
However, upon the multitude of products available on the market, which one is recommended? Depending on the surface, the type of cleaning agents and the type of testing (qualitative or quantitative), they are all suitable choices.
It is critical to verify that the chosen neutralizer has an efficient action against the used sanitizers. The above chart represents the most effective equivalence amongst the cleaning agents and the most common neutralizers.
For instance, if a quantitative method is to be used, it is very important to consider a neutralizing agent which doesn’t support the bacterial growth such as the neutralizing buffer.
Finally the versatility of the sponge makes it a very popular choice. In fact, they are used for sampling equipment surfaces, floors, walls, work benches and even carcasses. They enable the sampling of large surfaces and the detection of lower levels of contamination at a lower cost of operation. To summarize, environmental sampling is an important tool to verify sources of contamination and adequacy of sanitation process helping to refine the frequency and intensity of cleaning and sanitation, identifying “hot spots”, validate food safety programs, and provide early warning of trends that may need corrective action. Over all, it provides the firm conviction that products being manufactured are made under sanitary conditions.
Gabriela Martinez is Scientific Director at Labplas. She can be reached at gmartinez@labplas.com.
 BioLab Business Magazine Together, we reach farther into the Canadian Science community
BioLab Business Magazine Together, we reach farther into the Canadian Science community